Generally speaking, a biological pacemaker is one or more types of cellular components that, when “implanted or injected into certain regions of the heart,” produce specific electrical stimuli that mimic that of the body’s natural pacemaker cells. It’s indicated for issues such as heart block, slow heart rate, and asynchronous heart ventricle contractions.
In healthy individuals, the sinoatrial node (SAN) is responsible for initiating excitation >100 000 times per day. As with all cardiac pacemaking cells in the specialized conduction system, SAN automaticity is driven by diastolic depolarization via several redundant pathways to ensure that the heart beats. Key cellular mechanisms include the hyperpolarization-activated inward current (If) and the Ca clock (inward Na/Ca exchange current activated by sarcoplasmic reticulum Ca2+ release).1 However, molecular, histological, electrophysiological, and in silico studies have revealed that the SAN is extremely complex, and that the redundant systems not only provide safety of pacemaking, but also allow precise modulation of heart rate to accommodate changing physiological demands.
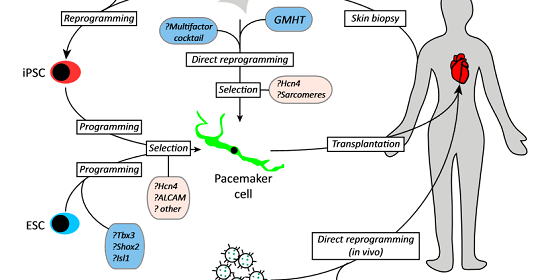
One of the most exciting recent developments with regard to biological pacemakers has been the exploration of human induced pluripotent stem cell–derived cardiomyocytes (iPSC-CMs) as potential pacemaking cells. With this approach, human somatic cells such as hair follicles or skin cells are reprogrammed to become pluripotent stem cells and are differentiated into cardiomyocytes.6 This unique approach circumvents many of the ethical issues associated with the use of human embryonic stem cell–derived cardiomyocytes (hESC-CMs) and allows the creation of pacemaker cells from a patient’s own tissue, making this approach immunocompatible.
Genetically engineered pacemakers could be a possible alternative to implantable electronic devices for the treatment of bradyarrhythmias. The strategies include upregulation of beta adrenergic receptors, conversion of myocytes into pacemaker cells and stem cell therapy. Pacemaker activity in adult ventricular myocytes is normally repressed by the inward rectifier potassium current (IK1). The IK1 current is encoded by the Kir2 gene family. Use of a negative construct that suppresses current when expressed with wild-type Kir2.1 is an experimental approach for genesis of genetic pacemaker. Hyperpolarisation activated cyclic nucleotide gated (HCN) channels which generate If current, the pacemaker current of heart can be delivered to heart by using stem cell therapy approach and viral vectors. The unresolved issues include longevity and stability of pacemaker genes, limitations involved in adenoviral and stem cell therapy and creation of genetic pacemakers which can compete with the electronic units.
Researchers Develop First Minimally Invasive Gene Therapy Procedure to Treat Heart Rhythm Disorders by Transforming Ordinary Heart Muscle Cells into Specialized Rhythm-Keeping Cells, Potentially Eliminating Future Need for Electronic Pacemakers
Cardiologists at the Cedars-Sinai Heart Institute have developed a minimally invasive gene transplant procedure that changes unspecialized heart cells into “biological pacemaker” cells that keep the heart steadily beating.
The laboratory animal research, published online and in today’s print edition of the peer-reviewed journal Science Translational Medicine, is the result of a dozen years of research with the goal of developing biological treatments for patients with heart rhythm disorders who currently are treated with surgically implanted pacemakers. In the United States, an estimated 300,000 patients receive pacemakers every year.
“We have been able, for the first time, to create a biological pacemaker using minimally invasive methods and to show that the biological pacemaker supports the demands of daily life,” said Eduardo Marbán, MD, PhD, director of the Cedars-Sinai Heart Institute, who led the research team. “We also are the first to reprogram a heart cell in a living animal in order to effectively cure a disease.”
These laboratory findings could lead to clinical trials for humans who have heart rhythm disorders but who suffer side effects, such as infection of the leads that connect the device to the heart, from implanted mechanical pacemakers.
Eugenio Cingolani, MD, the director of the Heart Institute’s Cardiogenetics-Familial Arrhythmia Clinic who worked with Marbán on biological pacemaker research team, said that in the future, pacemaker cells also could help infants born with congenital heart block.
“Babies still in the womb cannot have a pacemaker, but we hope to work with fetal medicine specialists to create a life-saving catheter-based treatment for infants diagnosed with congenital heart block,” Cingolani said. “It is possible that one day, we might be able to save lives by replacing hardware with an injection of genes.”
“This work by Dr. Marbán and his team heralds a new era of gene therapy, in which genes are used not only to correct a deficiency disorder, but to actually turn one kind of cell into another type,” said Shlomo Melmed, dean of the Cedars-Sinai faculty and the Helene A. and Philip E. Hixson Distinguished Chair in Investigative Medicine.
In the study, laboratory pigs with complete heart block were injected with the gene called TBX18 during a minimally invasive catheter procedure. On the second day after the gene was delivered to the animals’ hearts, pigs who received the gene had significantly faster heartbeats than pigs who did not receive the gene. The stronger heartbeat persisted for the duration of the 14-day study.
“Originally, we thought that biological pacemaker cells could be a temporary bridge therapy for patients who had an infection in the implanted pacemaker area,” Marbán said. “These results show us that with more research, we might be able to develop a long-lasting biological treatment for patients.”
Credit : Cedar-Sinai